Titration Curve of Strong Acid against Weak Base
(image)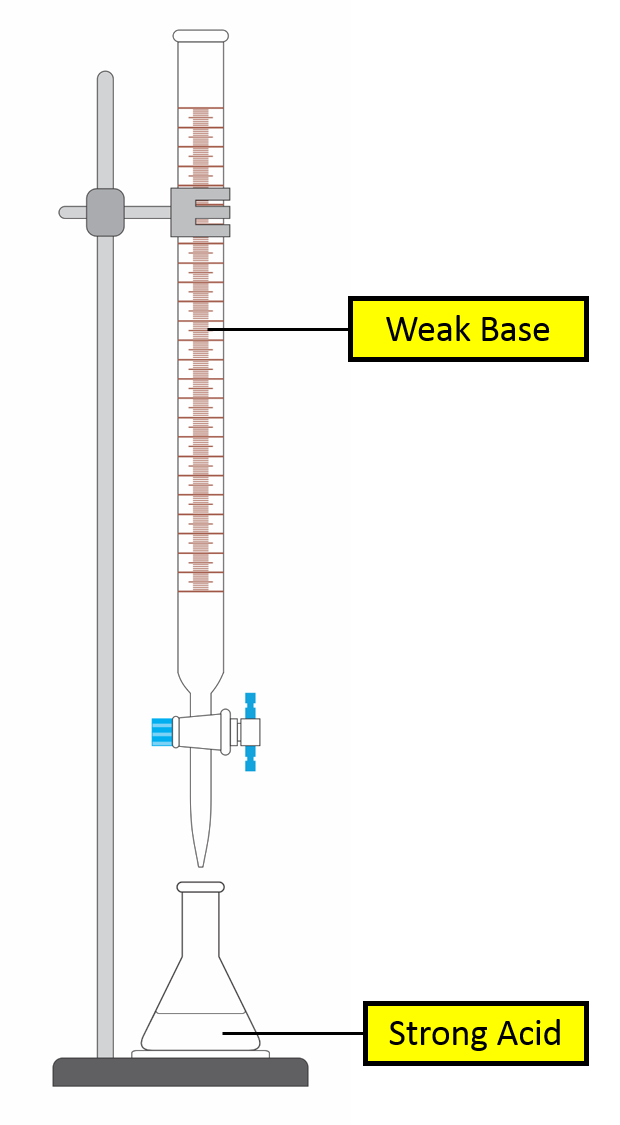
Titration Curve of Strong Acid against Weak Base
(image)
|
Self Check 1. Select a point by clicking the graph:Burette Volume pH 2. Identify the species present in the conical flask at that point: SA conj acid of WB xs WB 3. Is solution a buffer? yes |
|
||||||||||||||||||||||||||||||||||||||||||||||||||