About
Topics
- Atomic Bonding
- Chemistry
- Dipole
- Interaction Potential
- Molecules
Description
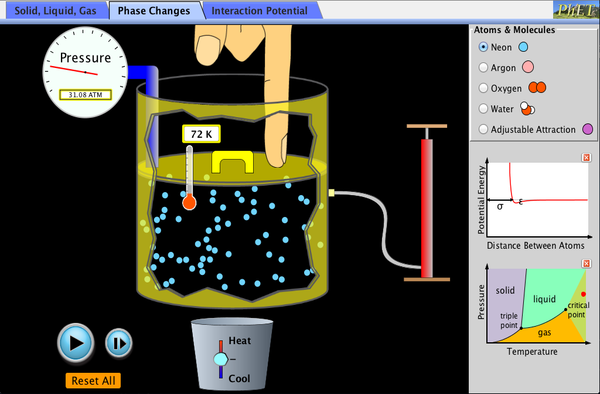
Watch different types of molecules form a solid, liquid, or gas. Add or remove heat and watch the phase change. Change the temperature or volume of a container and see a pressure-temperature diagram respond in real time. Relate the interaction potential to the forces between molecules.
Sample Learning Goals
- Describe a molecular model for solids, liquids, and gases.
- Extend this model to phase changes.
- Describe how heating or cooling changes the behavior of the molecules.
- Describe how changing the volume can affect temperature, pressure, and state.
- Relate a pressure-temperature diagram to the behavior of molecules.
- Interpret graphs of interatomic potential.
- Describe how forces on atoms relate to the interaction potential.
- Describe the physical meaning of the parameters in the Lennard-Jones potential, and how this relates to the molecule behavior.
For Teachers
need some CSI worksheerts?
Software Requirements
| Windows | Macintosh | Linux |
|---|---|---|
|
Microsoft Windows
XP/Vista/7
Sun Java 1.5.0_15 or later
|
OS 10.5 or later
Sun Java 1.5.0_19 or later
|
Sun Java 1.5.0_15 or later |
Credits
taken from https://phet.colorado.edu/en/simulation/states-of-matter-basics
| Design Team | Third-party Libraries | Thanks To |
|---|---|---|
|
|


