About
Adding Heat
Introduction
The purpose of this simulation is to show how the temperature of water changes over time, as it undergoes phase transitions, when supplied with energy at a constant rate.Description of simulation
This simluation features both a graph of temperature over time, as well as a 'world view' to facillitate the students' understanding of how the shape of the water changes as it undergoes various phase transitions.Translations
| Code | Language | Translator | Run | |
|---|---|---|---|---|
 |
||||
Software Requirements
| Android | iOS | Windows | MacOS | |
| with best with | Chrome | Chrome | Chrome | Chrome |
| support full-screen? | Yes. Chrome/Opera No. Firefox/ Samsung Internet | Not yet | Yes | Yes |
| cannot work on | some mobile browser that don't understand JavaScript such as..... | cannot work on Internet Explorer 9 and below |
Credits
Shaun Quek; lookang; inspired by a version from Andrew Duffy; http://physics.bu.edu/~duffy/HTML5/heat_addHeat.html
end faq
Sample Learning Goals
[text]
For Teachers
Adding Heat to Water 2021 JavaScript HTML5 Applet Simulation Model
For the simulation, click here.
Description of the model
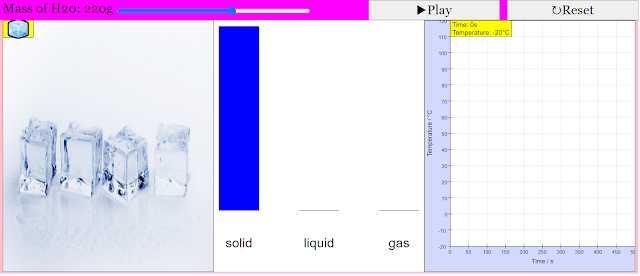
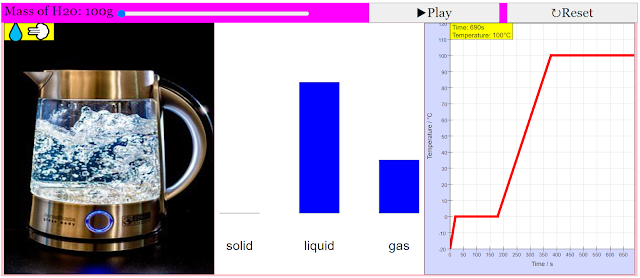
This model attempts to describe a sample of water that initially starts at -20°C, and has heat added to it at a constant rate. By default, the total mass of water is 100g. However this can be altered using the slider at the top right, to a maximum of 300g.
This simulation shows 3 panels: real world (for illustration purposes), a bar chart for the mass of water in the solid, liquid and gas states, and a graph of temperature as a function of time.
When the simulation is started, the temperature of water increases with time. However, as the water transitions between states (from sold to liquid, and liquid to gas), the temperature of water remains constant with time. The bar chart would also illustrate how the mass of water changes between states.
With these numbers and the information from the graph, you should be able to figure out the rate at which energy is added to the water sample. Don't forget to be amazed by how much time the sample spends changing phase.
Research
[text]
Video
[text]
Version:
Other Resources
http://physics.bu.edu/~duffy/HTML5/heat_addHeat.html
end faq
Facebook Social Comments


.png
)